 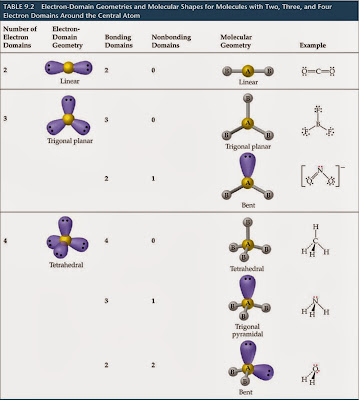
The rest of the octet will be completed with lone pairs. For this purpose, hydrogen is treated as an exception hydrogen requires two valence electrons to fulfill the octet rule.Īnother couple of useful guidelines are that, for many non-metal elements, the number of covalent bonds is equal to (8-, as listed in the table below. This is called the octet rule, because the final number of valence electrons around most atoms is eight.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
